 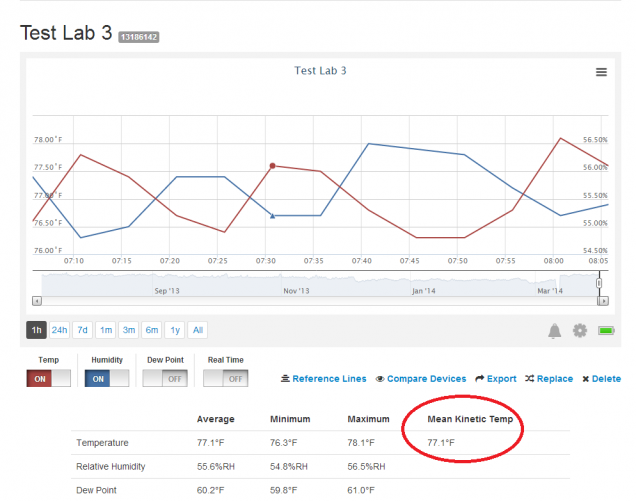
Similarly, the gas pressure is higher if the average velocity of molecules is higher. We gain a better understanding of pressure and temperature from the kinetic theory of gases, which assumes that atoms and molecules are in continuous random motion.īecause a huge number of molecules will collide with the wall in a short time, we observe an average force per unit area. Using the molecular mass of nitrogen latextextN2latex from the periodic table. My approach, assuming you are starting with a column of temperatures, would be: 1) calculate k at each temperature in an adjacent column 2) use the AVERAGE() function on this column to get the average k 3) Algebraically rearrange the Arrhenius equation to get T at k, and use that formula in Excel to get the mean kinetic temperature.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
